Phase I
Phase I Safety Review — NTX-101
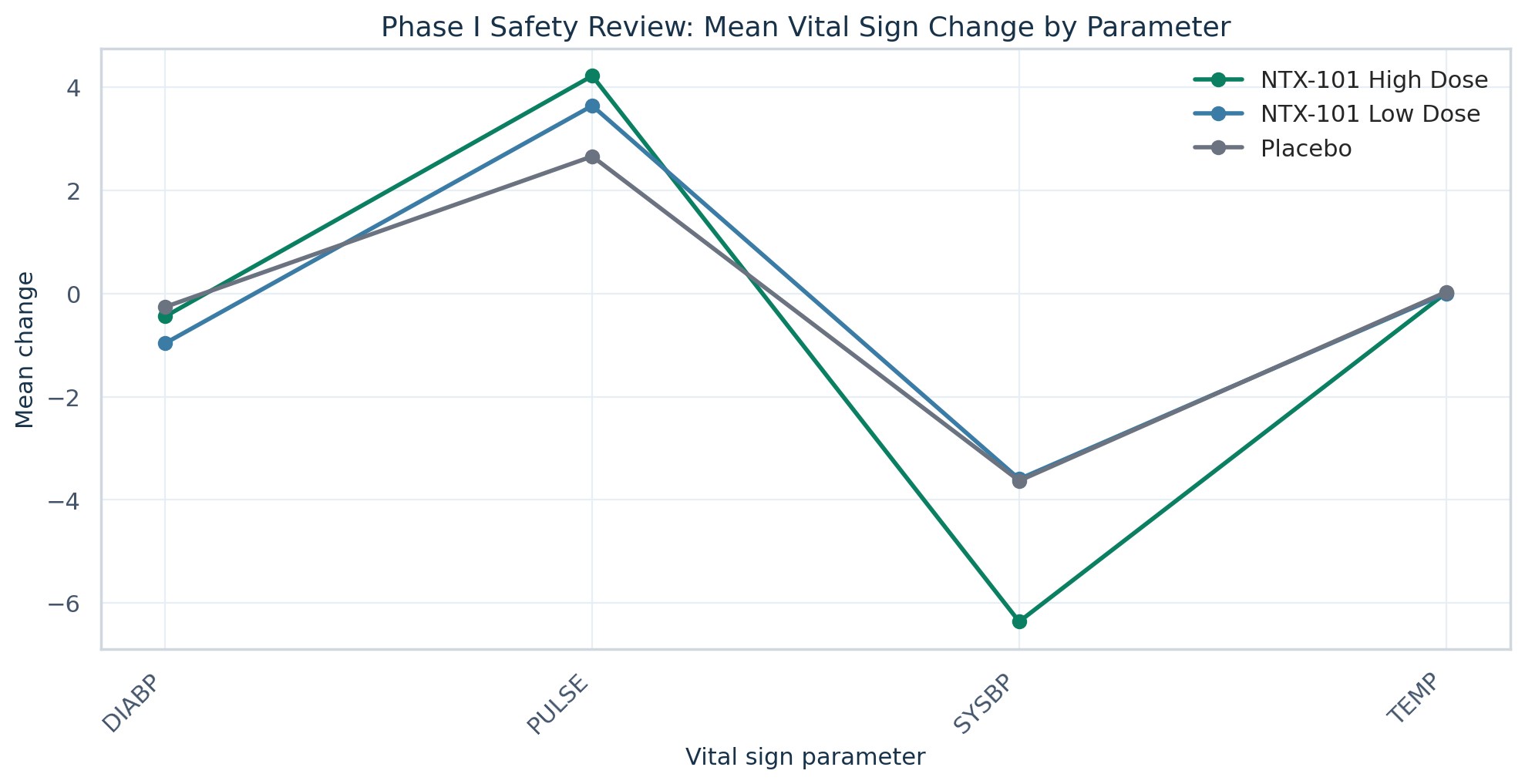
Safety, tolerability, laboratory, and vital-sign review for NTX-101 early development assessment.
Gate Decision: GO
Key safety metrics
| Metric | Value |
|---|
| AE rate difference vs placebo (pp) | 13.82 |
|---|
| Any AE p-value | 0.0057 |
|---|
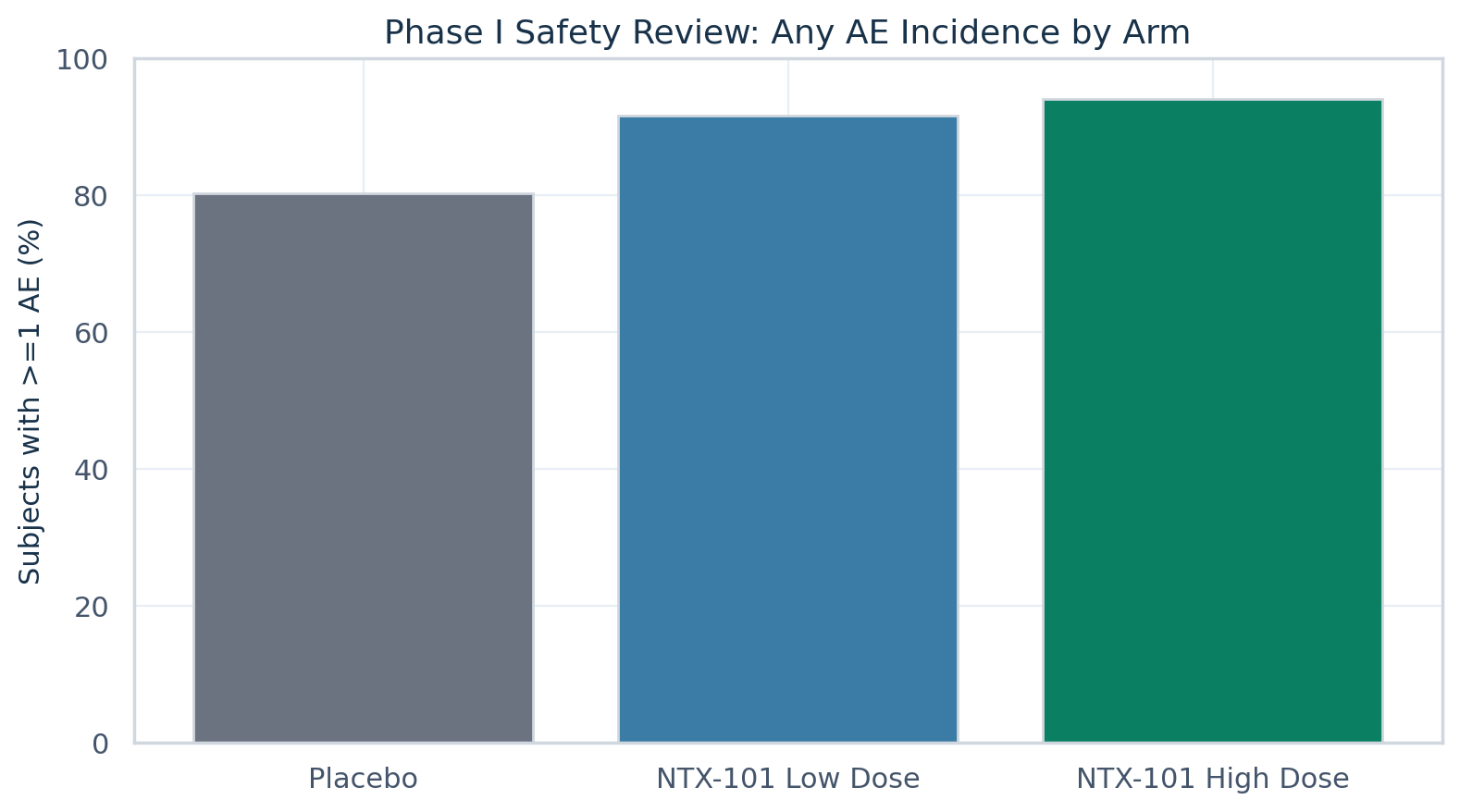
| Active any-AE rate (%) | 92.86 |
|---|
| Placebo any-AE rate (%) | 80.23 |
|---|
| Serious AE rate difference vs placebo (pp) | 2.38 |
|---|
| Serious AE p-value | 0.553 |
|---|
| Active serious-AE rate (%) | 1.79 |
|---|
| Placebo serious-AE rate (%) | 0.0 |
|---|
| Critical lab cluster | False |
|---|
| Treatment-related death imbalance | False |
|---|
Population summary
| Treatment | Subjects | Mean age |
|---|
| ntx101_high | 84 | 74.38 |
| ntx101_low | 84 | 75.67 |
| placebo | 86 | 75.21 |
AE incidence
| Treatment | N subjects | Subjects with AE | AE rate (%) |
|---|
| ntx101_high |
84 |
79 |
94.05 |
| ntx101_low |
84 |
77 |
91.67 |
| placebo |
86 |
69 |
80.23 |
Statistical checks
| Comparison | Statistic | p-value |
|---|
| Age balance active vs placebo | -0.1661 | 0.8683 |
| Any AE rate active vs placebo | 13.82 | 0.0057 |
| Serious AE rate active vs placebo | 2.381 | 0.553 |
Figures
![]()